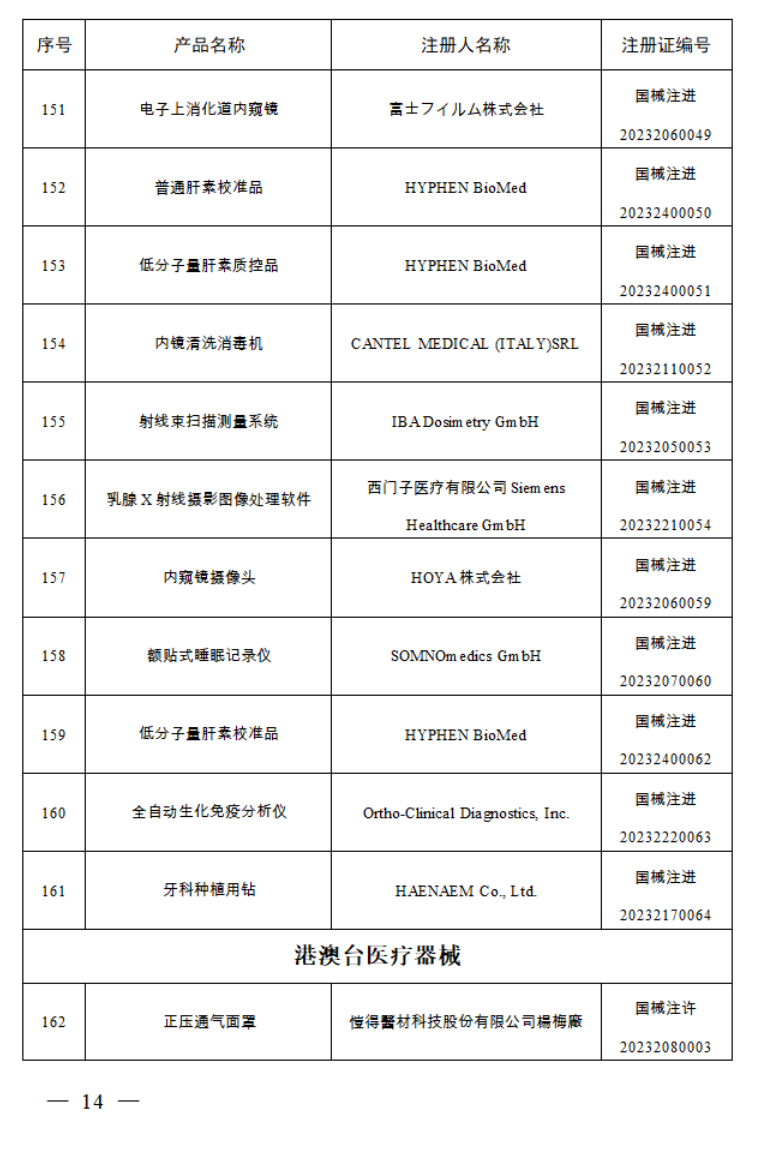
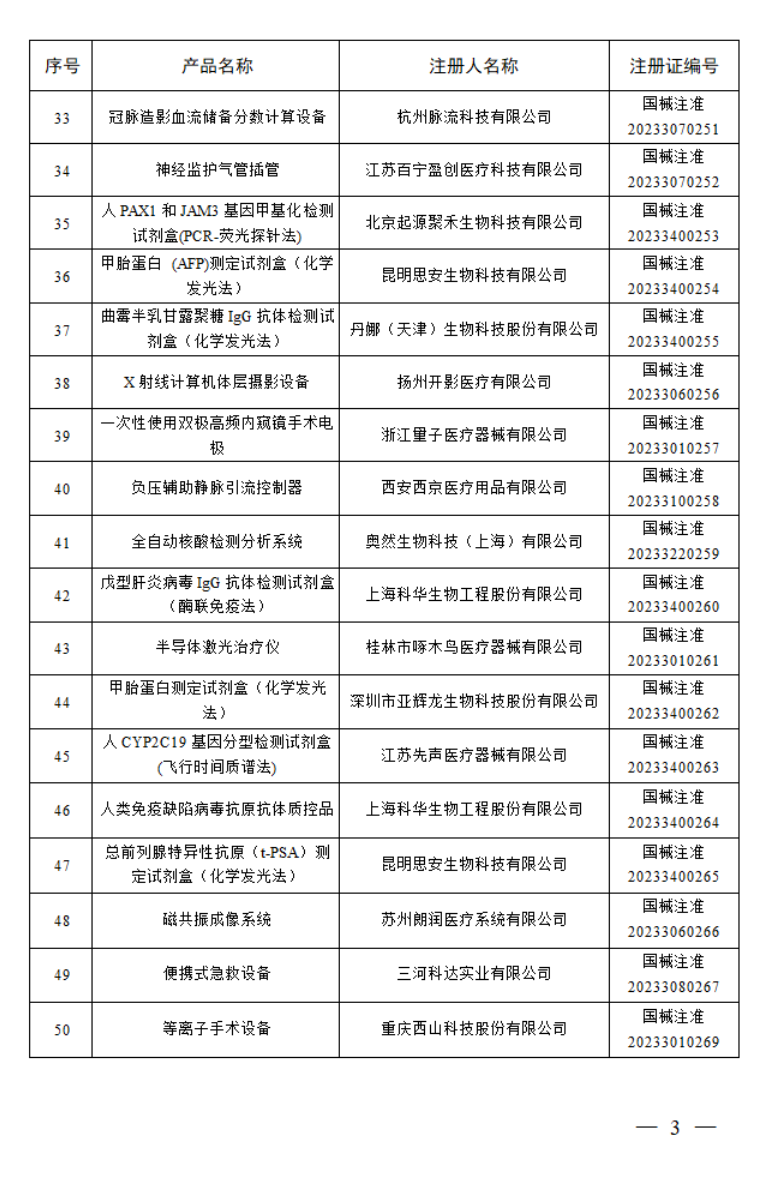
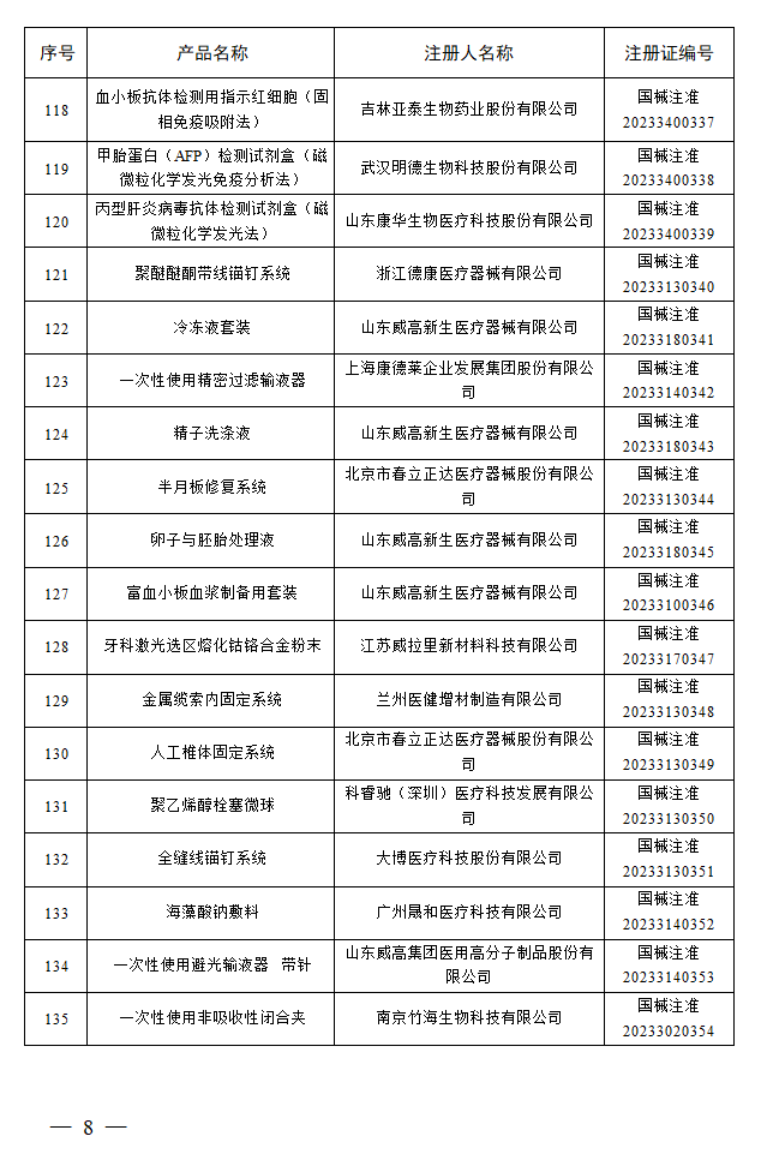
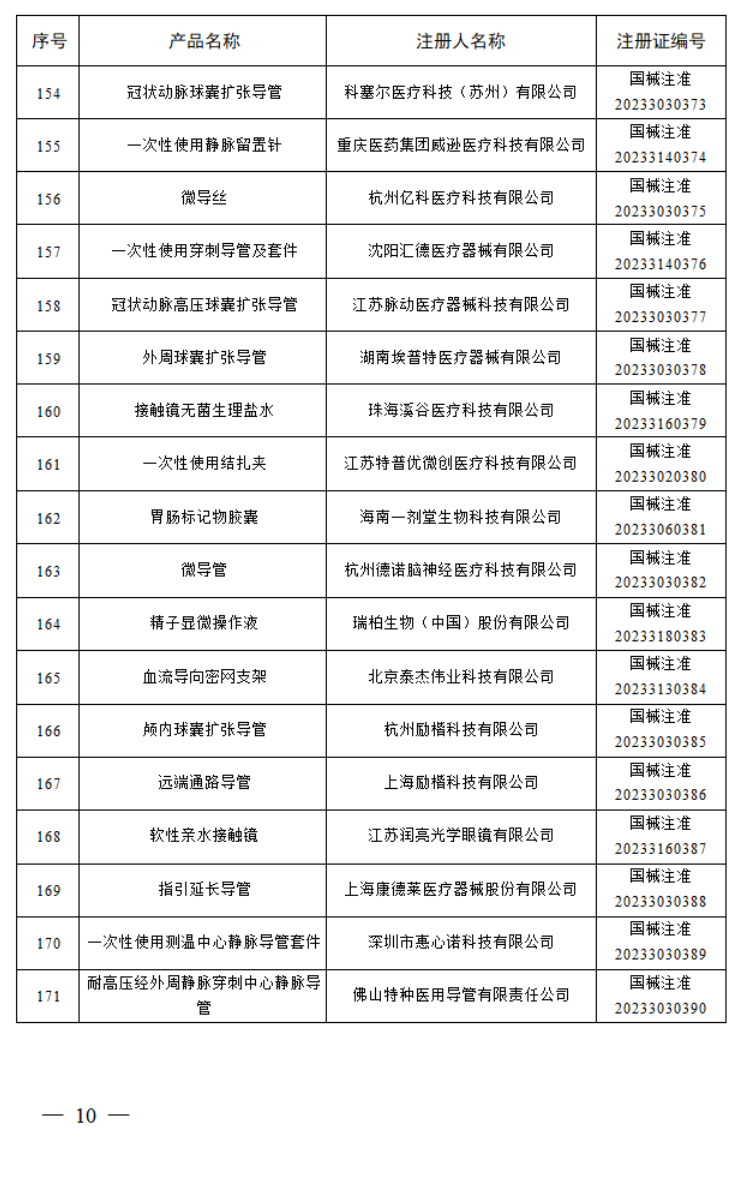
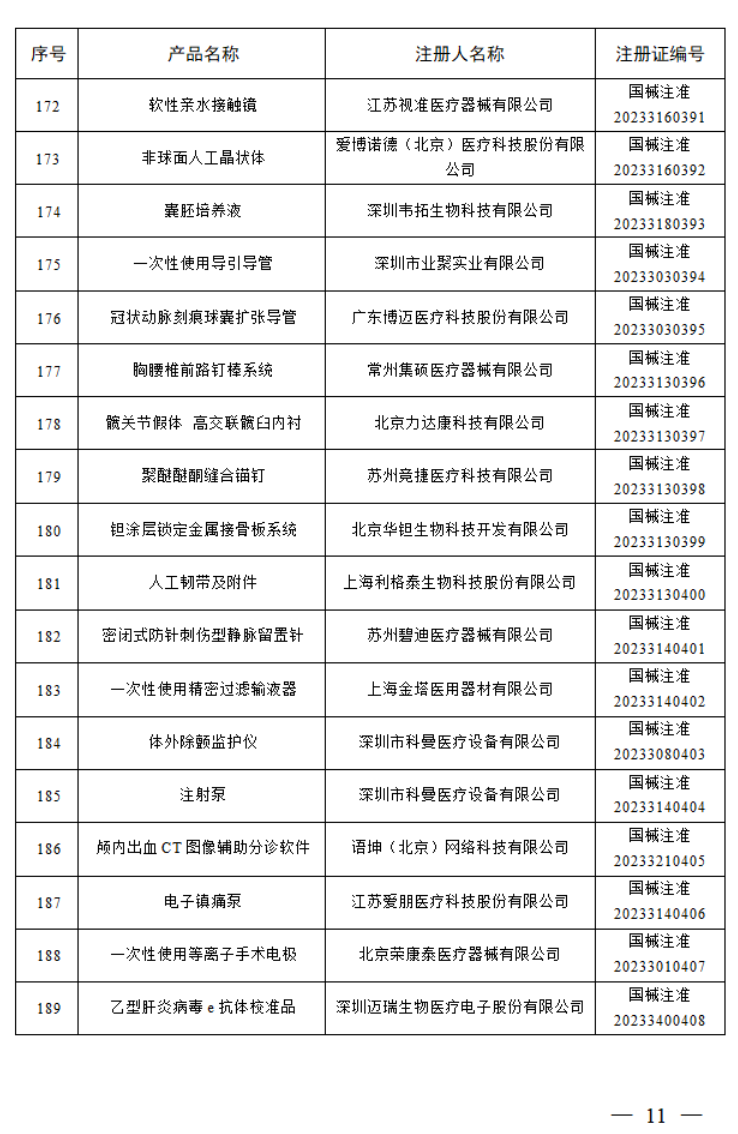
2023年7月,国家药监局共批准注册医疗器械产品197个。其中,境内第三类医疗器械产品150个,进口第三类医疗器械产品21个,进口第二类医疗器械产品21个,港澳台医疗器械产品5个。
据统计,今年1-7月,国家药监局共批准注册医疗器械产品1392个。批准注册医疗器械产品目录如下:

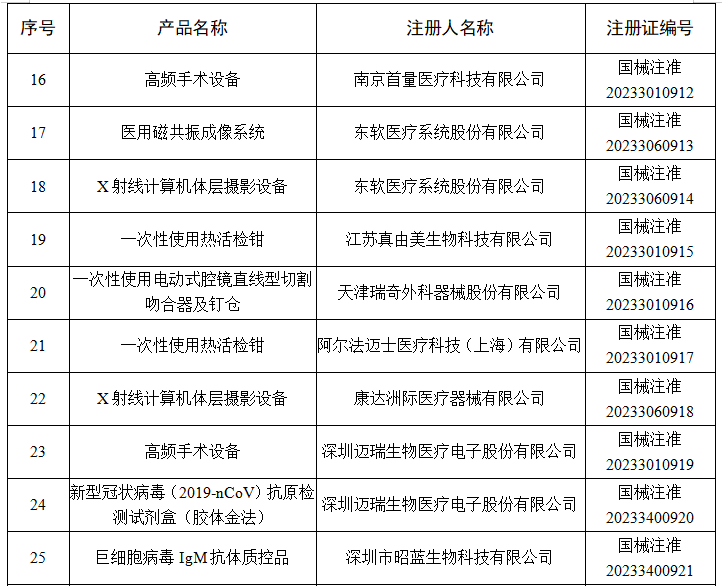


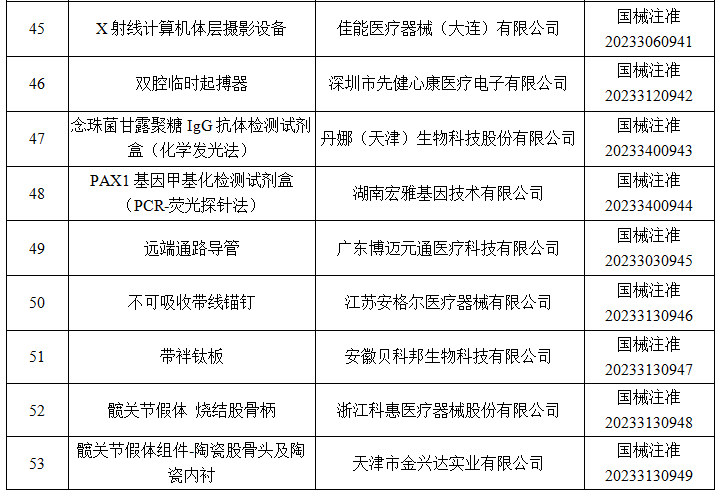
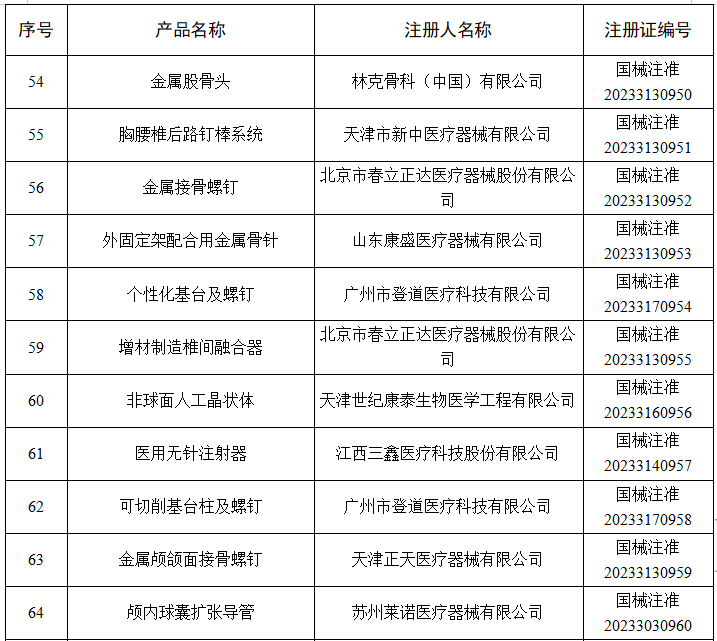









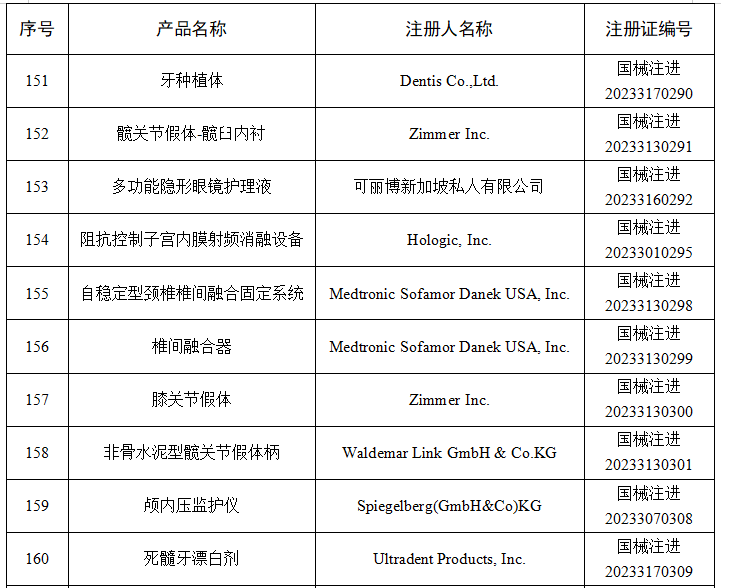




6月
2023年6月,国家药监局共批准注册医疗器械产品264个。其中,境内第三类医疗器械产品198个,进口第三类医疗器械产品46个,进口第二类医疗器械产品19个,港澳台医疗器械产品1个。





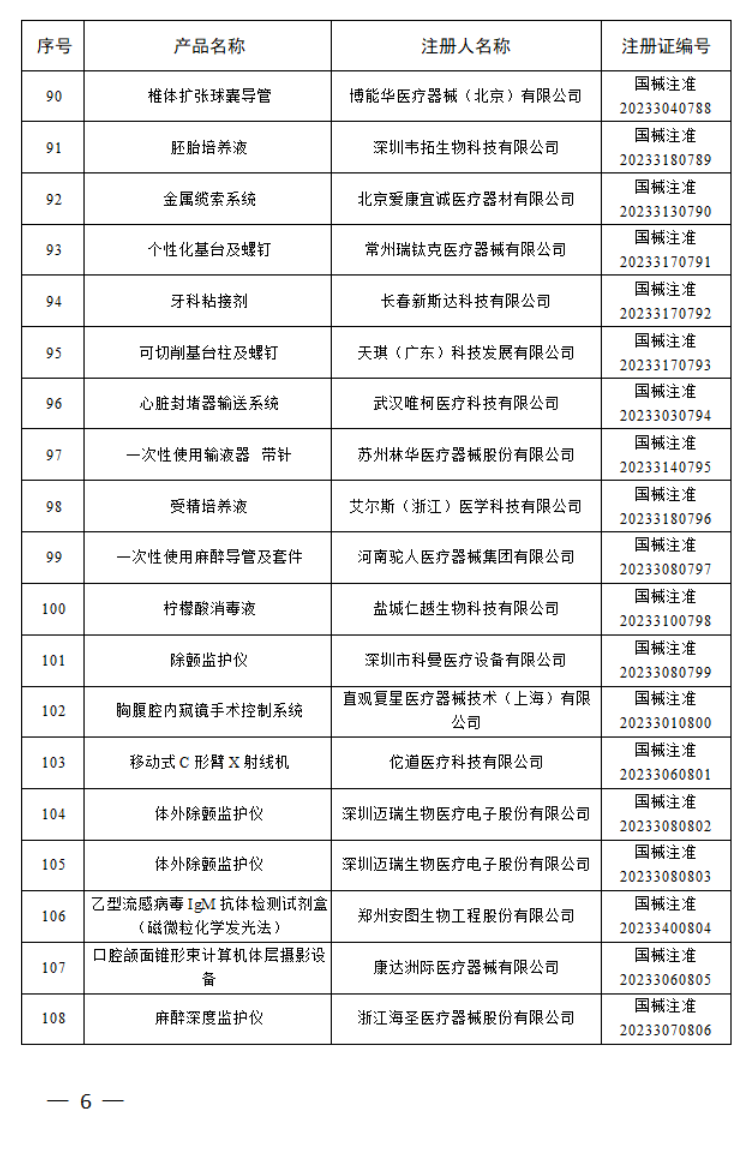
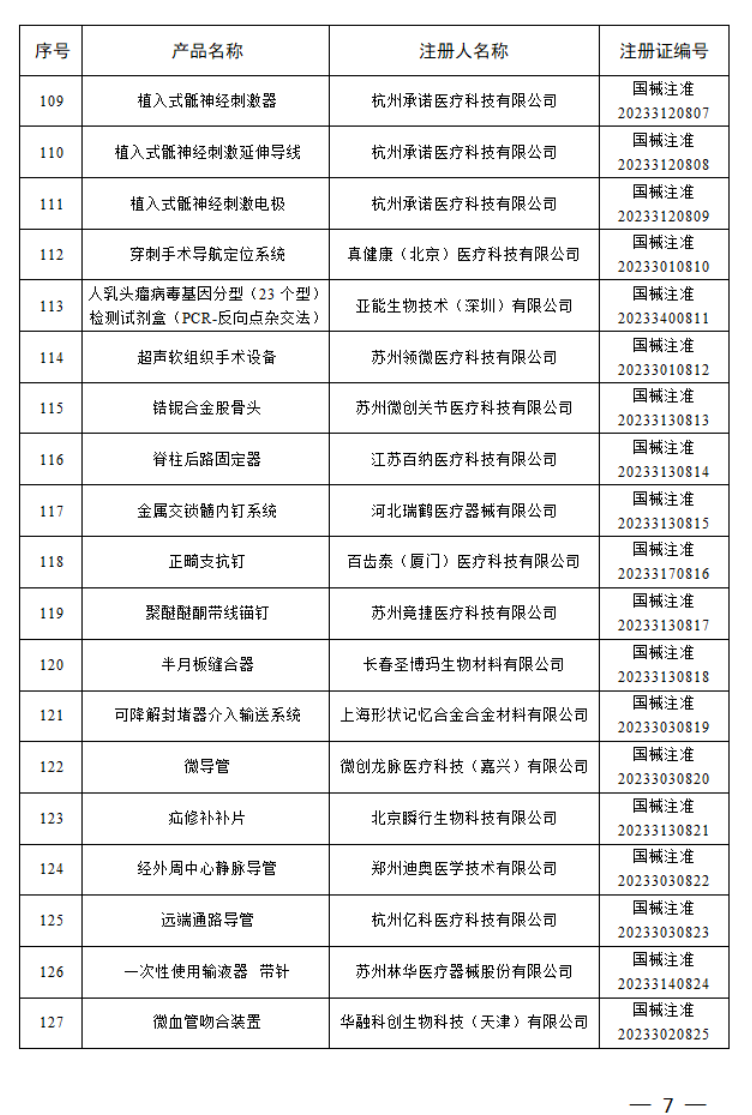








2023年5月,国家药监局共批准注册医疗器械产品175个。其中,境内第三类医疗器械产品128个,进口第三类医疗器械产品27个,进口第二类医疗器械产品20个。

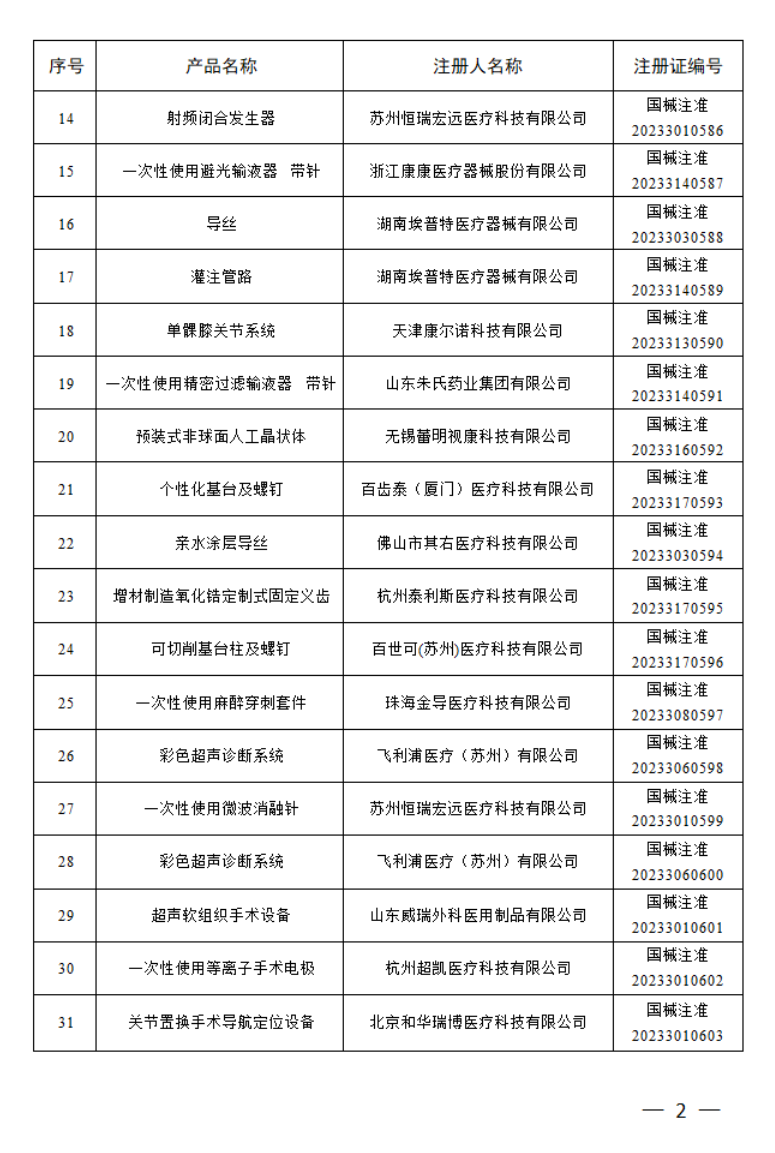

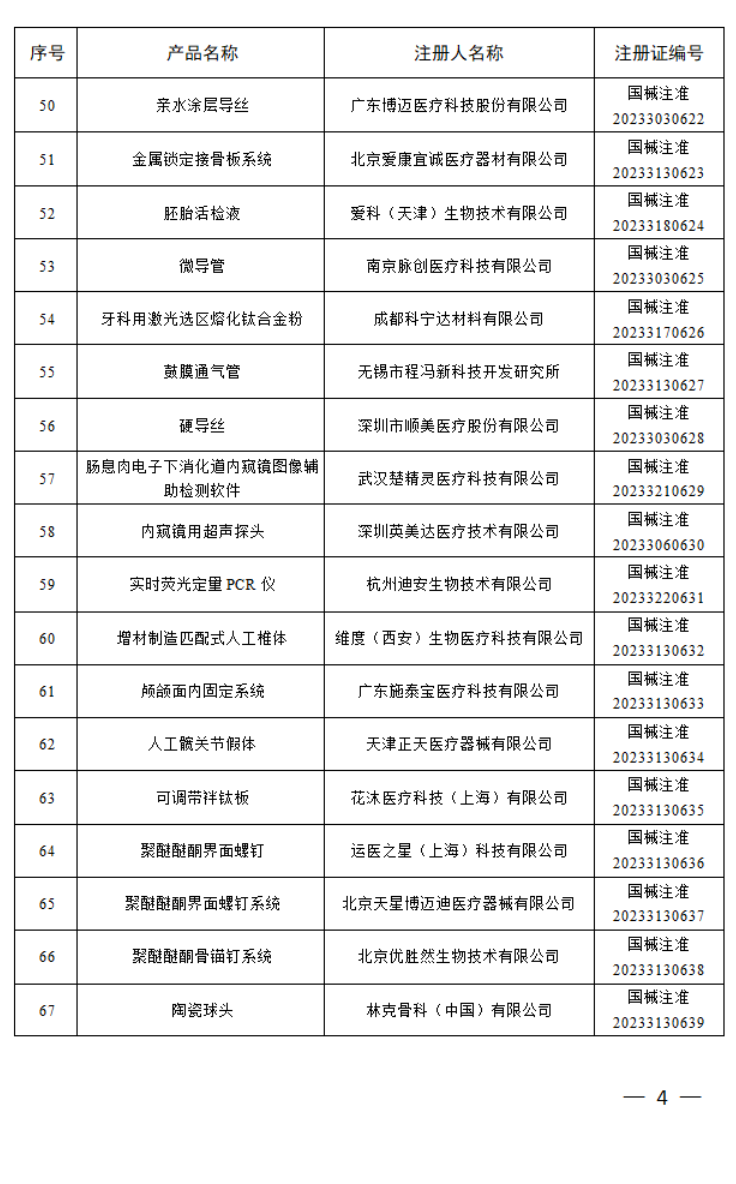


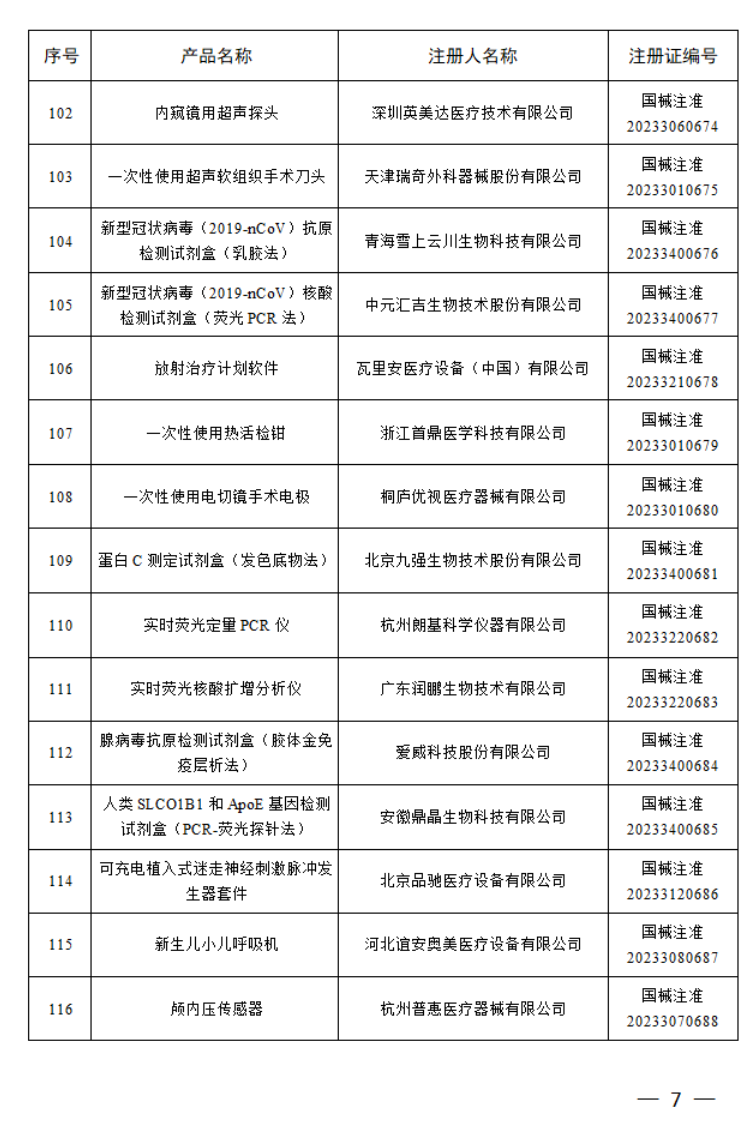




4月
2023年4月,国家药监局共批准注册医疗器械产品180个。其中,境内第三类医疗器械产品125个,进口第三类医疗器械产品21个,进口第二类医疗器械产品33个,港澳台医疗器械产品1个。






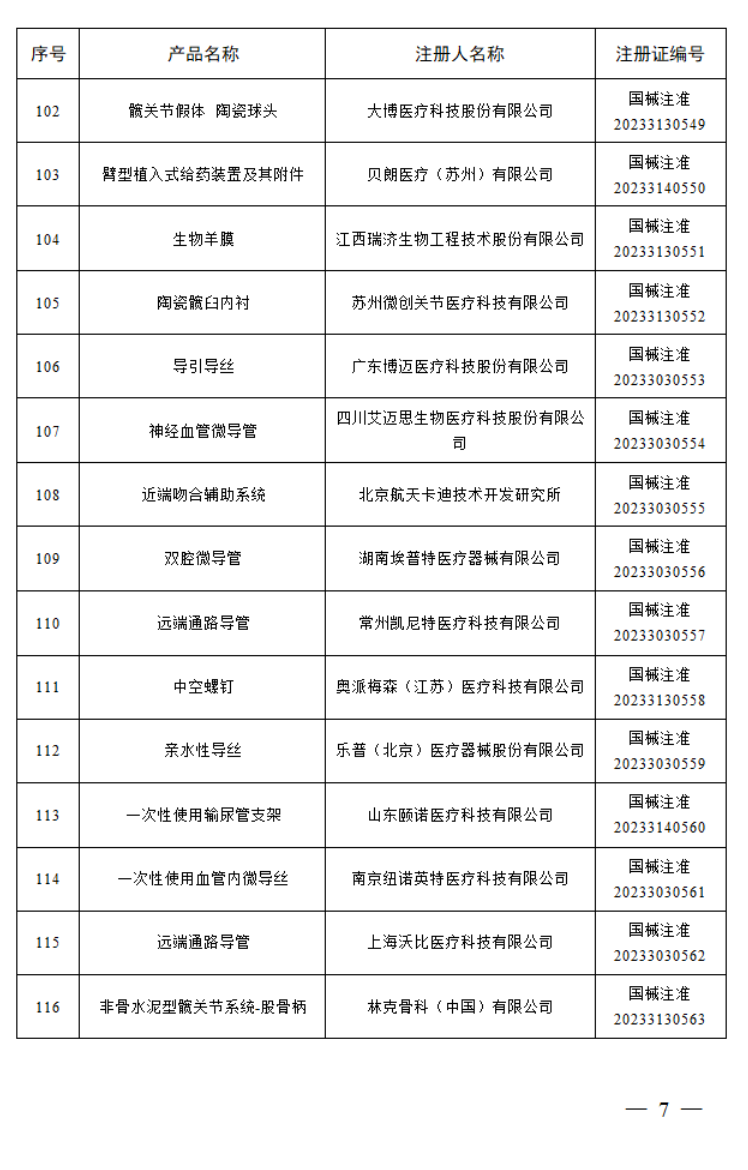




3月
2023年3月,国家药监局共批准注册医疗器械产品290个。其中,境内第三类医疗器械产品227个,进口第三类医疗器械产品33个,进口第二类医疗器械产品26个,港澳台医疗器械产品4个。

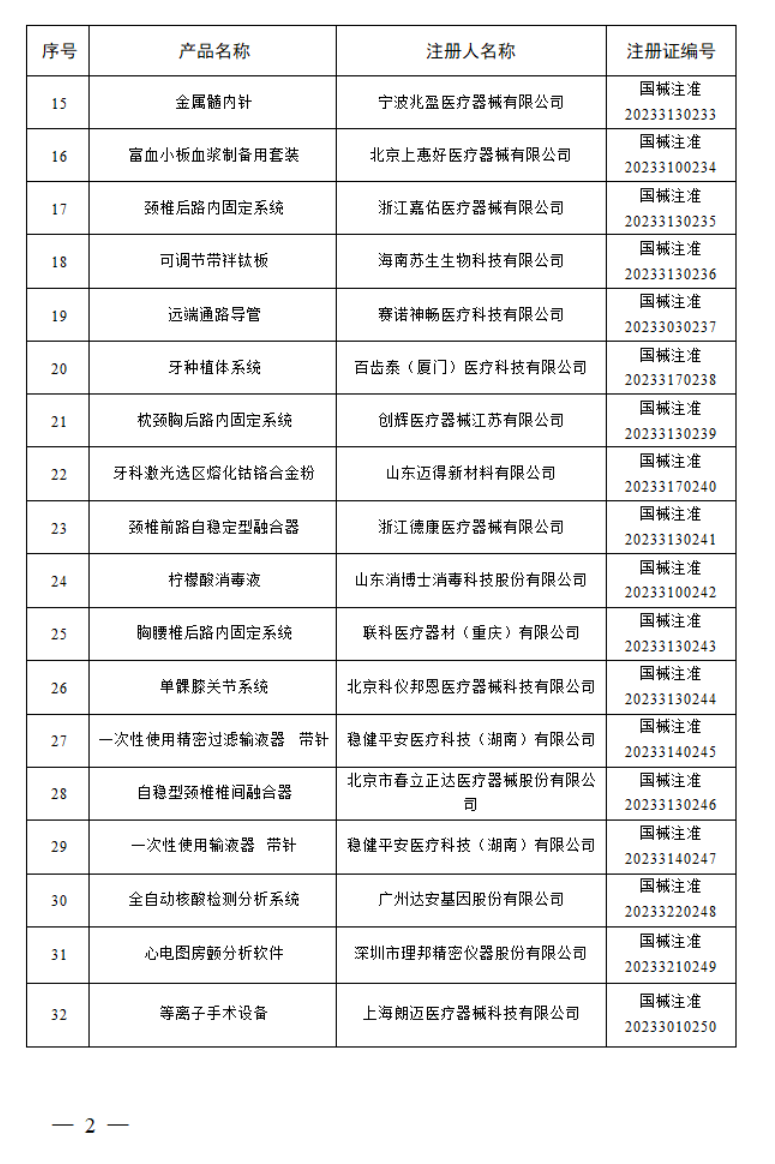









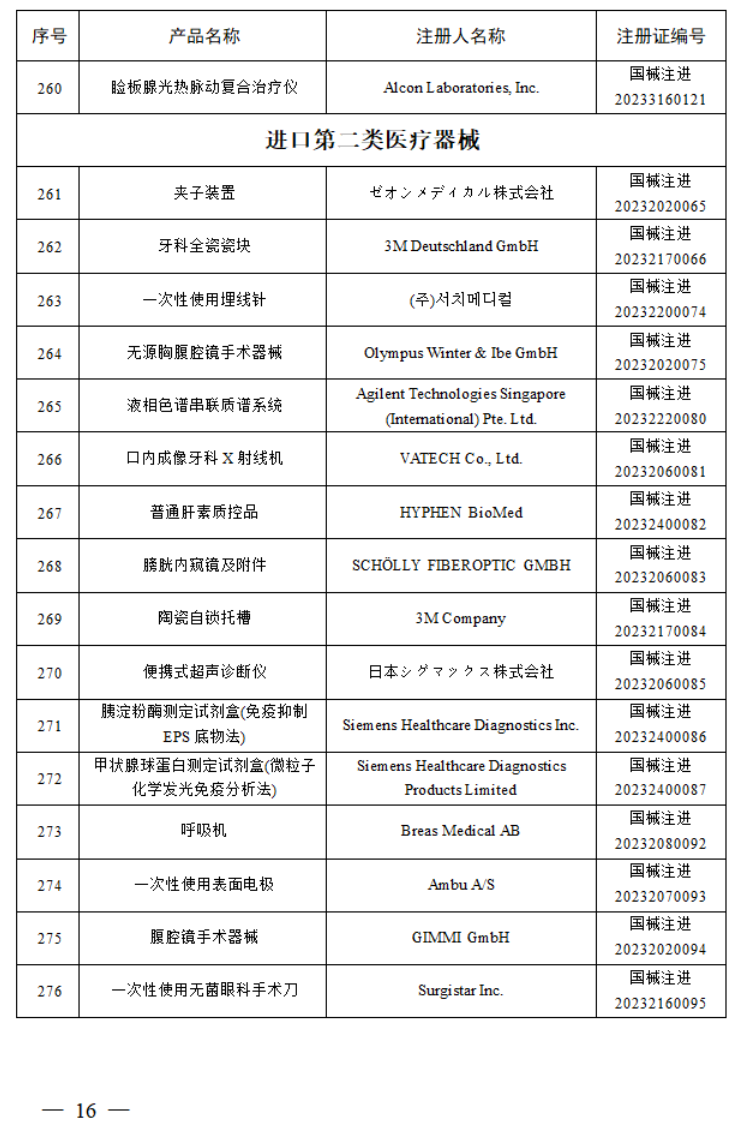
2023年2月,国家药监局共批准注册医疗器械产品162个。其中,境内第三类医疗器械产品120个,进口第三类医疗器械产品19个,进口第二类医疗器械产品22个,港澳台医疗器械产品1个。
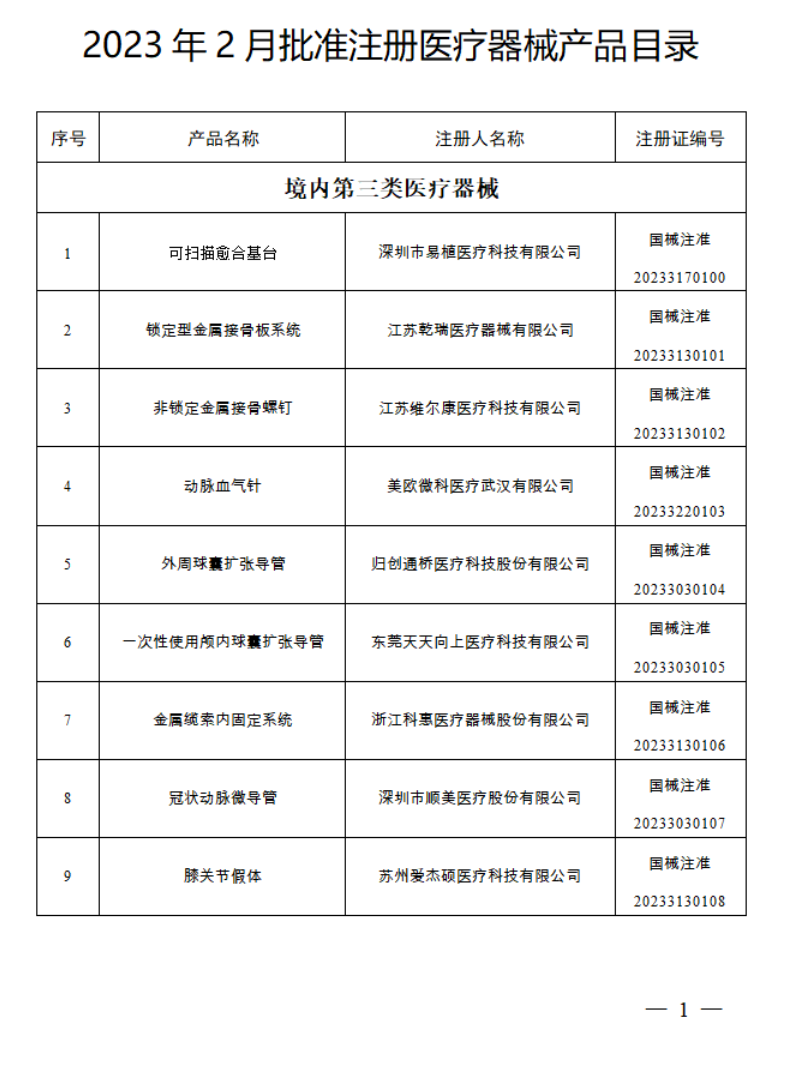







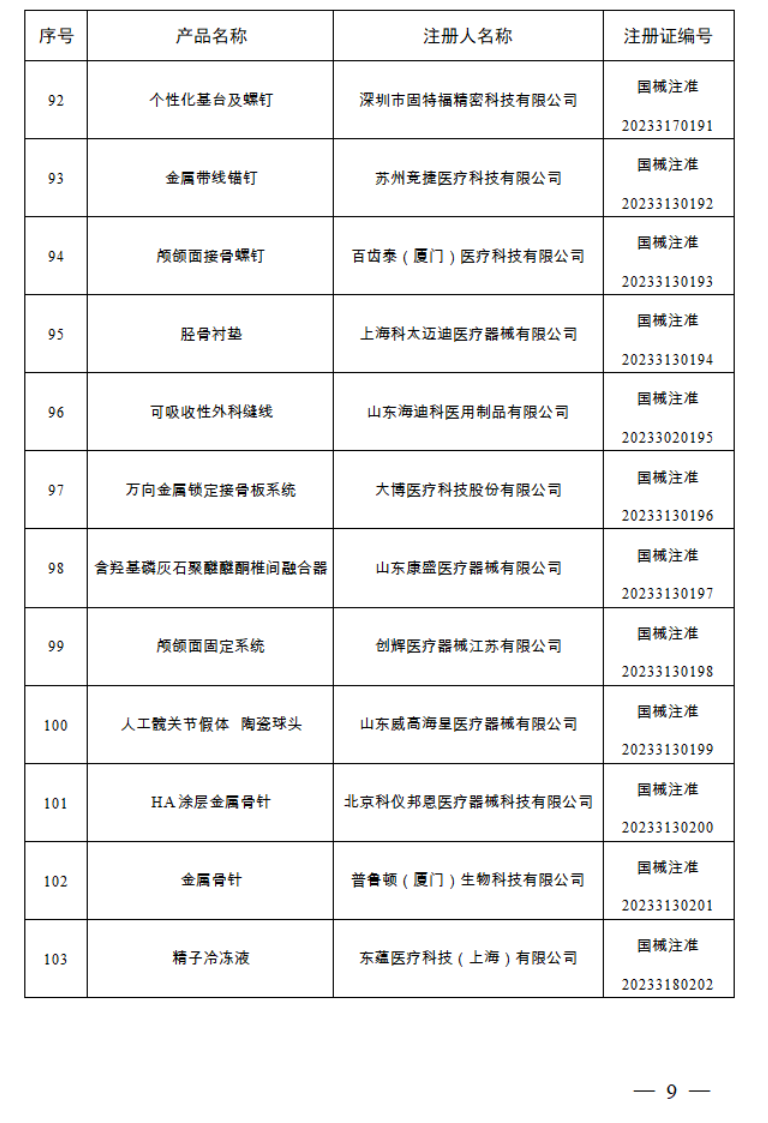

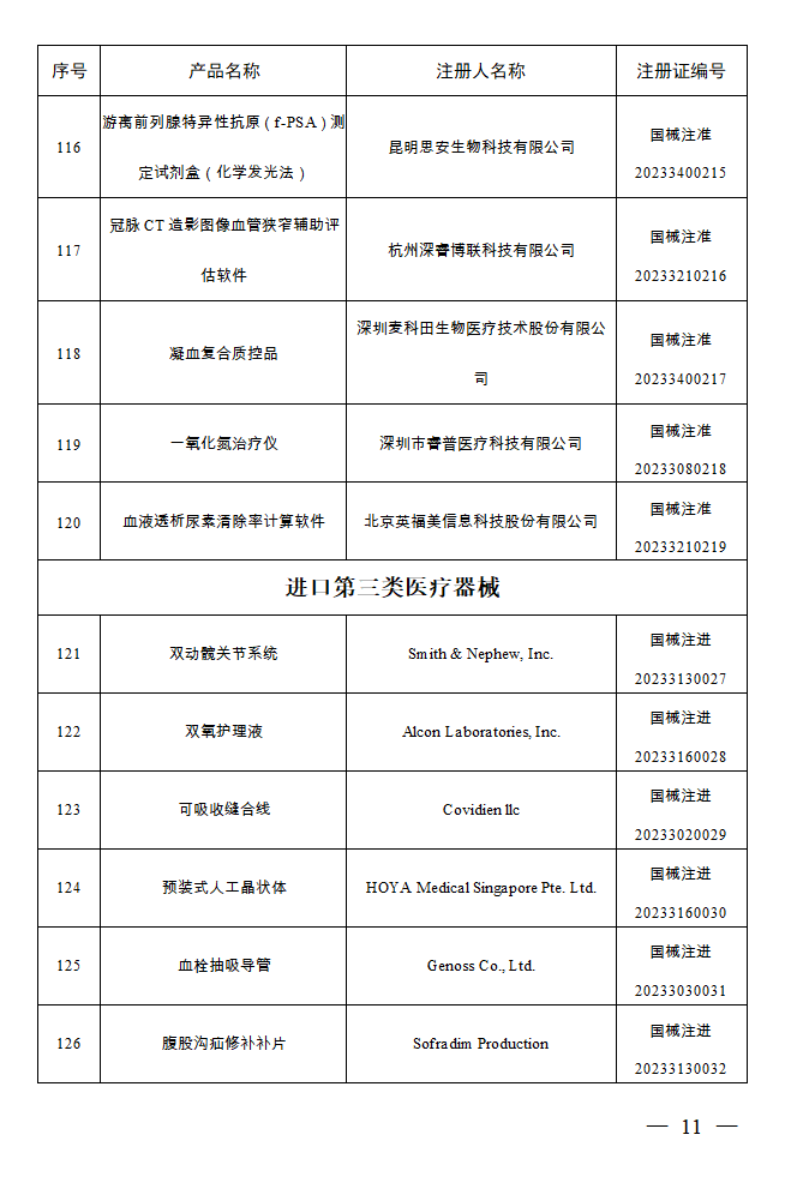


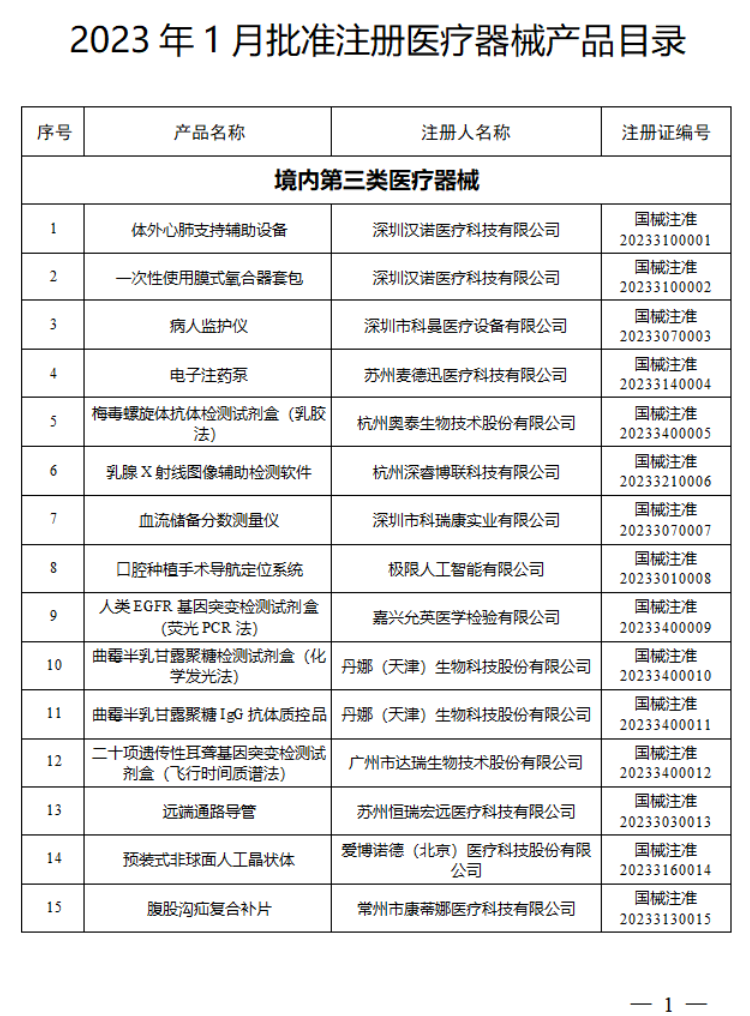
2023年1月,国家药监局共批准注册医疗器械产品124个。其中,境内第三类医疗器械产品99个,进口第三类医疗器械产品11个,进口第二类医疗器械产品12个,港澳台医疗器械产品2个。






本文著作权属原创者所有,不代表本站立场。我们转载此文出于传播更多资讯之目的,如涉著作权事宜请联系删除。